Pharmaceutical Automation

Industrial automation solutions for pharmaceutical manufacturing environments, designed to support compliance, data integrity, and reliable production.
At Stratos Control Systems, we deliver standards-based automation systems that help pharmaceutical manufacturers maintain validation, improve traceability, and reduce operational risk.
From PLC programming and SCADA systems to control panel design and system upgrades, our solutions are built to meet the demands of regulated environments.
Pharmaceutical Automation FAQs
How is automation used in pharmaceutical manufacturing?
Automation in pharmaceutical manufacturing controls processes such as dosing, mixing, and packaging. It ensures precision, repeatability, and compliance with strict regulatory standards.
What systems are used in pharmaceutical automation?
Pharmaceutical automation uses PLC systems, SCADA platforms, HMIs, and validated control systems to monitor and control production processes.
How does automation support compliance in pharmaceuticals?
Automation supports compliance through data logging, audit trails, and controlled processes, helping manufacturers meet regulatory requirements and maintain product quality.
Pharmaceutical Automation Challenges
Pharmaceutical manufacturing environments present complex automation challenges, including:
-
Validation and compliance requirements (GAMP-aligned approaches)
-
Ageing or unsupported PLC and SCADA systems
-
Poor documentation and unclear system ownership
-
Controlled upgrades in live production environments
-
Cleanroom and utility system integration
-
Data visibility and traceability requirements
By addressing these challenges at the control system level, we help reduce risk and simplify ongoing compliance.
How We Support Pharmaceutical Manufacturers

.webp)
Control Panel Design (AutoCAD & E-Plan)
We design pharmaceutical-grade control panels using AutoCAD and E-Plan, focusing on clarity, standards compliance, and long-term support.
-
Standards-based panel layouts
-
Clear segregation and labelling
-
Designed for validation and audit
-
Fully documented electrical schematics
PLC & HMI Programming

We deliver structured, maintainable PLC and HMI software designed to support validation, controlled change, and long-term reliability.
-
PLC programming and upgrades
-
HMI development with consistent standards
-
Clear alarms and fault diagnostics
-
Software documentation and version control support
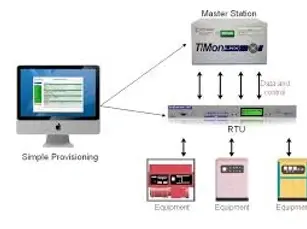
SCADA Systems for Pharmaceutical Environments
We design and integrate SCADA systems that support monitoring, traceability, and performance insight while maintaining data integrity.
-
SCADA system design and integration
-
Alarm management and audit-friendly reporting
-
Trends, batch data, and historical records
-
Platforms including Ignition, WinCC, FactoryTalk View SE
%20%26%20Utilities%20Integration%20Pharma.webp)
Building Management Systems (BMS) & Utilities Integration
Pharmaceutical facilities rely heavily on utilities and environmental control. We integrate BMS and utility systems to provide controlled, centralised visibility.
-
Cleanroom environmental monitoring
-
HVAC, utilities, and services integration
-
Alarm handling and reporting
-
Centralised operator interfaces

Control System Upgrades & Refurbishment
We deliver planned automation upgrades that minimise disruption while maintaining compliance.
-
Obsolete PLC and SCADA replacement
-
Control panel refurbishment
-
Phased upgrades and parallel systems
-
Structured testing and handover
Automation Platforms We Support
Stratos Control Systems primarily delivers pharmaceutical automation solutions using trusted industrial platforms:
-
Siemens
-
Allen Bradley
-
Mitsubishi
We also support legacy and specialist systems, particularly as part of upgrade and modernisation projects.

Automation Designed for Regulated Pharmaceutical Environments
Pharmaceutical automation systems must balance process control, data integrity, and regulatory compliance.
Legacy systems, poor documentation, or uncontrolled software changes increase validation effort and operational risk.
Stratos Control Systems delivers automation systems that are:
-
Structured and well documented
-
Designed for controlled change
-
Reliable and maintainable
-
Suitable for validation and audit
Our focus is on practical, compliant automation that supports both production and regulatory objectives.
Why Choose Stratos Control Systems
-
Experience working in regulated pharmaceutical environments
-
Standards-based design and documentation
-
Strong focus on validation, traceability, and controlled change
-
Expertise across PLC, HMI, SCADA, and BMS systems
-
Practical, risk-aware engineering approach
We deliver automation systems that are compliant, reliable, and supportable.
Frequently Asked Questions
Talk to Us About Pharmaceutical Automation
Whether you are upgrading legacy control systems, improving monitoring and traceability, or planning a new pharmaceutical automation project, Stratos Control Systems can help.